Orbital Diagram Example
Molecular orbital theory Orbital diagrams — overview & examples Orbitals bond valence theory bonding atomic covalent chemistry sp oxygen hybrid structure electron two hydrogen libretexts organic bonds mcc draw
Orbital Diagrams and Electron Configuration - Basic Introduction
Electron orbitals electronic quantum chemistry electrons numbers structure model atoms introductory orbital number figure arrangement atomic chem level libretexts chapter Orbital diagrams — overview & examples Introductory chemistry 1.0
Orbital electron diagrams monahan
Orbital orbitals shape 4f shapes atomic quantum numberOrbital diagrams to print Electron orbitals electrons quantum chemistry numbers electronic structure introductory model orbital atoms figure atomic arrangement number energy ball libretexts chapterOrbital electron diagrams configuration diagram potassium atom 2s configurations 1s 3s 2p ppt powerpoint presentation slideserve.
Orbital diagram electron diagrams configuration filling orbitals chemistry structure chem example first atomic arrows libretexts below atomsOrbital electron diagrams configuration chemistry practice problems basic Orbital diagrams3.7: electron arrangement- the quantum model.

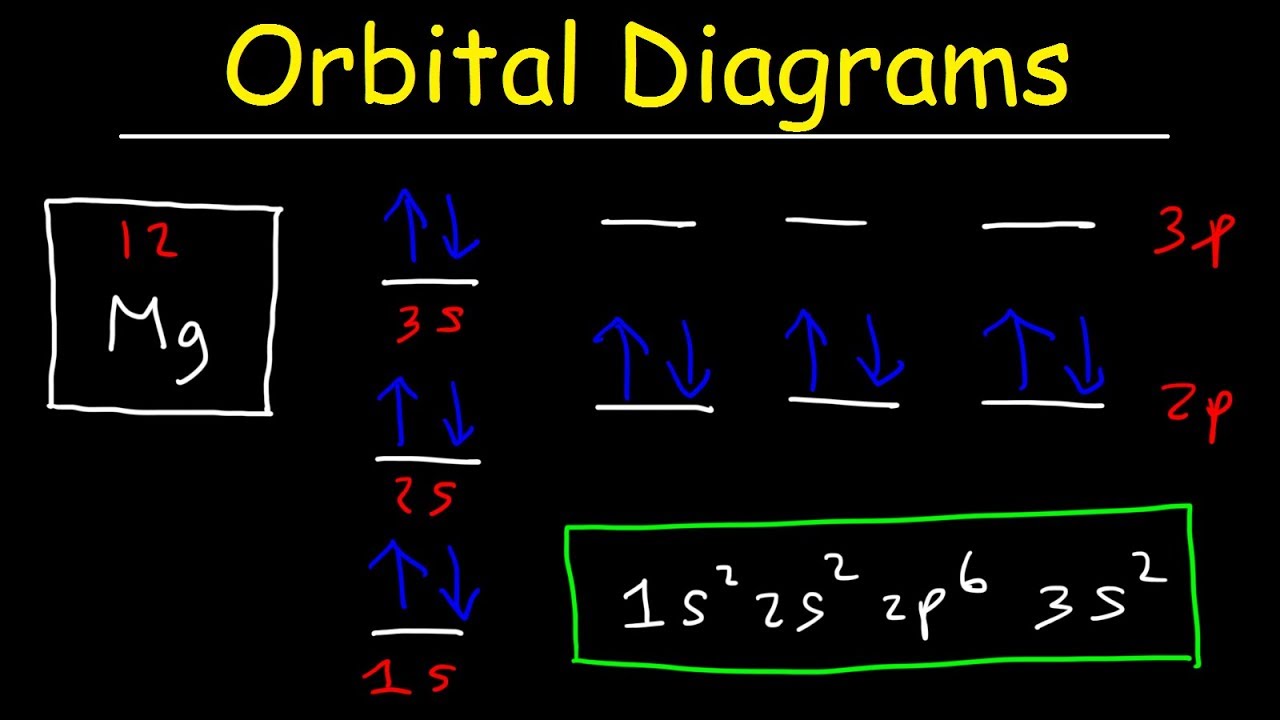
1.4: electron configuration and orbital diagrams
What is the shape of f-orbital??? + exampleOrbital orbitals pic2fly Orbital molecules diagram orbitals diatomic bonding chemistry of2 structure delocalized atomic electrons libretexts chem correlation valence geometry atoms stable homonuclear10.5: molecular orbital theory.
Electron orbitals electrons quantum numbers chemistry electronic structure introductory orbital model atoms figure atomic number arrangement libretexts ball energy chapterOrbital diagrams orbitals electrons monahan Figure8.6.electron orbitalsElectronic pairing structure orbital diagrams chemistry quantum diagram spin notation box electrons electron orbitals energy first boxes spins configurations level.

Orbitals atomic shapes chemistry chem cartesian atoms structure figure size space general
Molecular orbitals orbital bonding atomic pi atoms delocalized diatomic chem formation libretexts antibonding chemical combine molecules readings formed axis lobes1.7: atomic orbitals and covalent bonding 6.6: the shapes of atomic orbitalsOrbital diagrams and electron configuration.
.